2.6ü@ Crosses of mutants defective in the
sporophyte formation
Takako
Tanahashi
Introduction
Double
disruptants of PpLFY1*1 and PpLFY2*1
have drastically decreased rate of the sporophyte formation, less than 1%,
compared to that of the wild type, more than 90%.ü@ Most of the leaky sporophytes have aberrant morphology and spores
in those rarely germinate.ü@ On the other
hand, gametophores of the double disruptants have no phenotypic difference from
those of the wild type.ü@ This is a cross
method between the wild type and the PpLFY double disruptants
utilizing such characters of the latter.ü@
We obtained some sporophytes originated from cross-fertilization of
wild-type sperm and egg cells of PpLFY disruptants.ü@ They are distinguished from other
sporophytes formed on the double disruptants by their morphology and the
germination rate of containing spores.
Our method is
effective especially in the crosses of mutants defective in the sporophyte
formation for the following two reasons.
1. Maternal
strain is easily identified when crossing two strains with no morphological
difference in their gametophores.ü@
2. Jiffy-7
(medium used) increases the efficiency of the sporophyte formation.
ü@(*1 homologs of FLORICAULA/LEAFY genes in Physcomitrella
patens.ü@ Tanahashi et al.
Development 132 1727-1736 (2005))
Procedures
1) Inoculate
the wild-type and the double disruptant mosses (protonemata or gamtophores) on
a Jiffy-7, 3 cm in diameter before expansion.
2) Grow
mosses under long day condition (24L or 16L8D) at 25ºC
for 1-1.5 months and then under short day condition (8L16D) at 15ºC for 3 weeks in separate plastic boxes (Fig.
1A).ü@ Antheridia and archegonia
differentiate on gametophore shoot apices.
3) Check
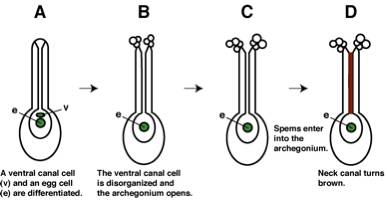
there are archegonia fully open and having uncolored neck canal yet (Fig. 2).
4) Place the wild-type and the disruptant mosses into a same
plastic box (Fig. 1B).
5) Submerge
these mosses with distilled water (Fig. 1C).ü@
Place it with gently shaking for 30 seconds, and then remove water above
mosses by decantation (Fig. 1D).
6) Continue
the culture under short day condition at 15ºC
for another 5 weeks.ü@ Collect
sporophytes formed on the double disruptant gametophores and examine genotypes
of progenies.
Result (all the
values are for colonies of PpLFY double disruptants)
|
ü@No. Jiffy-7 used. |
ü@ü@ü@ 71 |
|
ü@No. gametophores with archegoniaü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ |
ü@18019 |
|
ü@No. sporophytes* formed |
ü@ü@ 141 |
|
ü@No. sporophytes analyzed for their
genotypeü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ |
ü@ü@ü@ 87 |
|
ü@No.
sporophytes formed by cross-fertilizationü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ |
5 (normal
morphology, ü@ germination rate was good) |
|
ü@No.
sporophytes formed by self-fertilization or parthenogenesisü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ü@ |
82 (largely abnormal
morphology, germination rate was bad) |
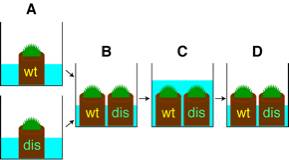
Fig.
1 Outline of crosses. (wt; wild type, dis; PpLFY
double disruptant)
Fig.
2 Development of an archegonium.ü@ Stage
C is proper for the manipulation.
Comments
We could not
obtain higher rate of sporophyte formation of PpLFY double
disruptants when performing cross-manipulation than when not performing it.
ü@ü@ % sporophyte formation (with
cross-manipulation)ü@ü@ü@ 0.78% (N = 18019)
ü@ü@ % sporophyte formation (without
cross-manipulation)ü@ 0.79% (N = 3152)
Apparently,
this procedure remains much to be improved.
ü@
For the
present, the most effective way for the success of cross is just;
1. Growing
healthy mosses
2. Performing
cross in a proper timing.
Therefore you
had better to prepare MANY mosses for your success.
The following
can increase the efficiency of cross-fertilization, though we have never tried
yet.
1. Repeating
submergence another several times. (If a sporophyte is not formed, new
archegonia will differentiate successively on a shoot apex.)
2. Condensing sperms with centrifugation and apply those
close to an archegonium.