| DIVISION OF BIO-ENVIRONMENTAL SCIENCE | ||||||||||
|
||||||||||
1) from Nihon Bioresearch Inc. |
||||||||||
Synthetic chemicals found in the environment
have the capacity to disrupt endocrine system development and function
in both wildlife and humans. This has drawn public concern since many
of these chemicals may bind to estrogen receptors (ER) and evoke estrogenic
effects. Early evidence that estrogenic chemicals could pose a threat
to human health during development came from studies of diethylstilbestrol
(DES), which was used to prevent premature birth and spontaneous abortion.
Laboratory experiments have demonstrated that exposure of animals
to sex hormones during perinatal life can cause permanent and irreversible
alterations of the endocrine and reproductive systems as well as the
immune system, nervous system, bone, muscle, and liver in both sexes.
Although many of these chemicals may bind to ER and evoke estrogenic
effects in wildlife and humans, the effects of estrogen are not well
understood even now. Thus, understanding the effects of sex hormones
at the molecular level, especially during development, is very important
to resolve these problems.
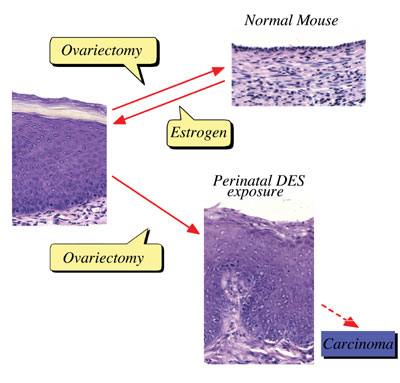
Perinatal sex-hormone exposure has been found to induce lesions in
reproductive tracts in female mice. The possible relevance of the
mouse findings to thedevelopment of cancer in humans has been emphasized.
In the early seventies, a close correlation between occurrence of
vaginal clear cell adenocarcinoma in young women and early intrauterine
exposure to DES was demonstrated. Many chemicals released into the
environment have the potential to disrupt endocrine function in wildlife
and humans. Some of these chemicals induce estrogenic activity by
binding to ER. The neonatal mouse model has been utilized especially
to demonstrate the long-term effects of early sex hormone exposure
on the female reproductive tract. Neonatal treatment of female mice
with estrogens induces various abnormalities of the reproductive tract:
ovary-independent cervicovaginal keratinization, adenosis, uterine
hypoplasia, epithelial metaplasia, oviductal tumors, polyovular follicles
(PF) and polyfollicular ovaries. Female reproductive tracts in mice
exposed prenatally to estrogen show altered expression of Hoxa genes
and Wnt genes and the analysis of knockout mice lacking Hoxa-10 or
Wnt7a show uterine hypoplasia. The growth response of neonatally DES-exposed
reproductive organs to estrogen is reduced, as are ER levels and epidermal
growth factor (EGF) receptor levels, in addition to other hormone
receptor levels.
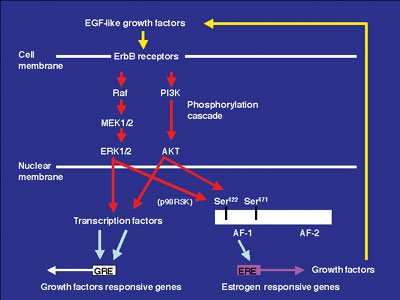
Growth factors and ER signaling cooperate to play essential roles in cell proliferation, differentiation and tumor progression in mouse reproductive organs. The mechanisms of the estrogen-dependent and -independent pathways remain unknown. EGFR and erbB2 were activated in the vaginal epithelium of mice by estrogen treatment. This activation was also encountered in vaginae from neonatally DES-exposed mice, along with the expression of EGF, TGF-a, HB-EGF, amphiregulin and neuregulin. Immunochitochemical analysis indicated that erbB2 was primarily expressed in vaginal epithelium. Serine 118 and 167 located in the AF-1 domain of ERa were phosphorylated in these vaginae. AG825, AG1478 or ICI 182,780, that are erbB2, EGFR and ER antagonists, respectively, administration blocked proliferation of vaginal epithelium induced by neonatal DES exposure. Signal transduction via EGFR and erbB2 could be related to the estrogen-induced vaginal changes and persistent erbBs phosphorylation and sustained expression of EGF-like growth factors, leading to ERa activation that may result in cancerous lesions in vaginae from neonatally DES- exposed mice later in life. To identify estrogen-responsive genes related to the proliferation and differentiation of mouse vaginal epithelial cells, we used differential display and identified a novel c-type lectin that encodes a membrane protein with a c-type lectin domain in the carboxyl-terminal region. Characterization of mRNA expression indicates that estrogen regulates the gene encodeing this novel c-type lectin in mouse vagina. Furthermore, this c-type lectin is found in epithelial cells, but not stromal cells, suggesting that it may be an important factor in the stratification and/or cornification of the vaginal epithelium of mice. We are continuing efforts to analyze its function during proliferation and differentiation in mouse vagina by estrogen treatment. Estrogenic compounds such as bisphenol A (BPA) and nonylphenol as
well as dioxins and PCBs were found in the human umbilical cord. BPA
can easily cross the placenta and enter the fetus in Japanese monkey
and mice. BPA can be found in fetal brain, testis and uterus when
given to pregnant mice and monkeys. Neonatal exposure to a high BPA
dose induced ovary-independent vaginal changes, polyovular follciles
and infertility lacking corpora lutera. Prenatal exposure to a low
BPA dose induced acceleration of vaginal opening in the offspring.
Thus, the developing mammal is sensitive to exposure to estrogenic
agents.
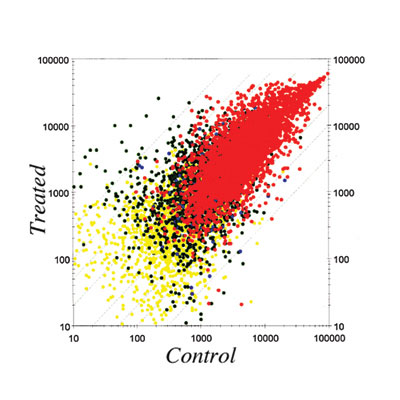
In order to clarify the molecular mechanisms of these effects, we
are studying changes in gene expression patterns induced by perinatal
exposure to chemicals or estrogen using differential display and DNA
microarray techiniques. We have found genes possibly related to the
ovary-independent changes by differential display. We also have clustered
groups of genes that are responsive to estrogenic stimuli in uterus
by using the DNA microarray system. We need to understand the molecular
background of the critical period during development, the low dose
effect of estrogenic chemicals and the molecular metabolism of hormones
and hormone-like agents in animals including humans. During embryogenesis, exogenous estrogen exposure induces abnormal
sex differentiation and the abnormal bone formation in African clawed
frog, Xenopus laevis, the cyprinodont fish, mummichog (Fundulus
heteroclitus), mosquitofish (Gambusia affinis). To analyze
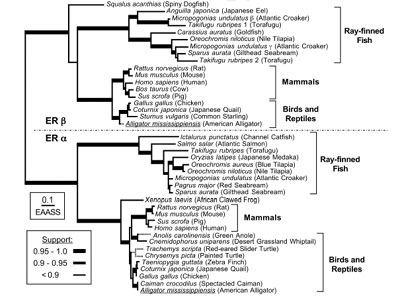
the function of estrogen, we have isolated cDNA clones of ERa
and b from F. heteroclitus, G. affinis.
In UK rivers exposure of roach (Rutilus rutilus – a
common cyprinid fish) to effluents from sewage treatment works, containing
complex mitures of endocrine disrupting chemicals (EDCs) has been
shown to alter sexual development and impact negatively on their reproductive
capabilities. To unravel the mechanisms of disruption of sexual development
in roach exposed to EDCs, we have isolated the cDNAs related to sex
determination and sexdifferentiation containing estrogen receptor,
aromatase, StAR, Sox9, vasa etc. We are examining the gene expression
during gonadal differentiation with or without EDCs exposure. Furthermore,
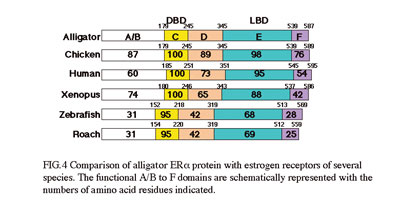
we have isolated cDNAs of steroid hormone receptors from American
alligators (Alligator mississippiensis). As the estrogen-responsive
genes must play important roles, we are isolating the estrogen-responsive
genes to understand the molecular physiology of estrogen action. Japanese
tree frog (Hyla japonica) takes water through ventral skin.
We found that sex steroids and endocrine disruptors interfere with
water absorption through ventral skin in frogs. Further, using the
amphibian and fish as model animals we aim to analyze the effects
of numerous chemicals released into the environment on endocrine system
function in wildlife.
Abnormalities caused by endocrine disrupting chemicals are reported
but the molecular mechanisms of the effects are not well studied.
Although estrogen receptor is one of the strongest candidates possibly
responsible for the endocrine disrupting function of many chemicals,
it alone cannot explain the variety of phenomena induced by endocrine
disrupting chemicals. Thus, we are also looking for new target molecules
that may be responsible for endocrine disruption. In parallel, we
also are studying the ligand-binding mechanisms of nuclear receptors
to hormones and other chemicals.
Publication List: Adachi, T., Matsuno, Y., Sugimura, A., Takano, K., Koh, K.B., Sakurai, K., Shibayama, T., Iguchi, T., Mori, C. and Komiyama, M. (2003) ADAM7 (a disintegrin and metalloprotease 7) mRNA is suppressed in mouse epididymis by neonatal exposure to Diethylstilbestrol. Mol Reprod Dev. 64, 414-421. Inui, M., Adachi, T., Takenaka, S., Inui, H., Nakazawa, M., Ueda, M., Watanabe, H., Mori, C., Iguchi, T. and Miyatake, K. (2003) Effect of UV screens and preservatives on vitellogenin and choriogenin production in male medaka (Oryzias latipes). Toxicology 194, 43-50. Kato, H., Ota, T., Furuhashi, T, Ohta, Y. and Iguchi T. (2003) Changes in reproductive organs of female rats treated with bisphenol A during the neonatal period. Reprod. Toxicol. 17, 283-288. Katsu, Y., Lubahn, D.B. and Iguchi T. (2003) Expression of a novel c-type lectin in the mouse vagina. Endocrinology 144, 2597-2605. Kohno, S., Kamishima, Y. and Iguchi, T. (2003) Molecular cloning of an anuran V (2) type [Arg (8) ] vasotocin receptor and mesotocin receptor: functional characterization and tissue expression in the Japanese tree frog (Hyla japonica). Gen. Comp. Endocrinol. 132, 485-498. Miyahara, M., Ishibashi, H., Inudo, M., Nishijima, H., Iguchi, T., Guillette, L.J. and Arizono, K. (2003) Estrogenic activity of a diet to estrogen receptors –alpha and –beta in an experimental animal. J. Health Sci. 49, 481-491. Nakahata, S., Kotani, T., Mita, K., Kawasaki, T., Katsu, Y., Nagahama, Y. and Yamashita, M. (2003) Involvement of Xenopus Pumilio in the translational regulation that is specific to cyclin B1 mRNA during oocyte maturation. Mech. Develop. 120, 865-880. Okada, A., Ohta, Y., Inoue, S., Hiroi, H., Muramatsu, M. and Iguchi, T. (2003) Expression of estrogen, progesterone and androgen receptors in the oviduct of developing, cycling and pre-implantation rats. J. Mol. Endocrinol. 30, 301-315. Sato, T., Fukazawa, Y., Kojima, H., Ohta, Y. and Iguchi, T. (2003) Multiple mechanisms are involved in apoptotic cell death in the mouse uterus and vagina after ovariectomy. Reprod. Toxicol. 17, 289-297. Tatarazako, N., Oda, S., Watanabe, H., Morita, M. and Iguchi, T. (2003) Juvenile hormone agonists affect the occurrence of male Daphnia. Chemosphere 53, 827-33. Tominaga, N., Ura, K., Kawakami, M., Kawaguchi, T., Kohra, S., Mitsui, Y., Iguchi, T. and Arizono, K. (2003) Caenorhabditis elegans responses to specific steroid hormones. J. Health Sci. 49, 28-33. Tominaga, N., Kohra, S., Iguchi, T. and Arizono, K. (2003) A multi-generation sublethal assay of phenols using the nematode Caenorhabditis elegans. J. Heath Sci. 49, 459-463. Urushitani, H., Nakai, M., Inanaga, H., Shimohigashi, Y., Shimizu, A., Katsu, Y. and Iguchi, T. (2003) Cloning and characterization of estrogen receptor alpha in mummichog, Fundulus hetetoclitus. Mol. Cell. Endocrinol. 203, 41-50. Watanabe H., Suzuki, A., Kobayashi, M., Lubahn D.B., Handa, H. and Iguchi, T. (2003) Similarities and differences in uterine gene expression patterns caused by treatment with physiological and non-physiological estrogens. J. Mol. Endocrinol. 31, 487-497. Watanabe H., Suzuki, A., Kobayashi, M., Takahashi, E., Itamoto, M., Lubahn D.B., Handa, H. and Iguchi, T. (2003) Analysis of temporal changes in the expression of estrogen-regulated genes in the uterus. J. Mol. Endocrinol. 30, 347-358. Adachi, T., Koh, K.B., Tainaka, H., Matsuno, Y., Ono, Y., Sakurai, K., Fukata, H., Iguchi, T., Komiyama, M. and Mori, C. (2004) Toxicogenomic difference between diethylstilbestrol and 17beta-estradiol in mouse testicular gene expression by neonatal exposure. Mol. Reprod. Dev. 67, 19-25. Katsu, Y., Bermudez, D.S., Braun, E., Helbing, C., Miyagawa, S., Gunderson, M.P., Kohno, S., Bryan, T.A., Guillette, L.J. and Iguchi, T. Molecular cloning of the estrogen and progesterone receptors of the American alligator. Gen. Comp. Endocrinol. (in press). Miyagawa, S., Katsu, Y., Watanabe, H. and Iguchi T. Estrogen-independent activation of erbBs signaling and estrogen receptor alpha in the mouse vagina exposed neonatally to diethylstilbestrol. Oncogene (in press) |